SVET101: An Introduction to the Scanning Vibrating Electrode Technique
Latest updated: November 18, 2024What is the Scanning Vibrating Electrode Technique?
The Scanning Vibrating Electrode Technique (SVET) measures the local electric field above the surface of a sample in solution, allowing electrochemical processes to be visualized as they occur. SVET has been widely applied in corrosion and coatings studies. It has also been applied to biological studies where it is known as Vibrating Probe.
In 1974 Jaffe and Nuccitelli introduced Vibrating Probe as a technique to visualize the small extracellular current associated with biological processes including growth and wound healing [1]. Vibrating Probe found enduring use as the Scanning Vibrating Electrode Technique in the closely related fields of corrosion and coatings starting in the 1980s, after introduction to the field by Hugh Isaacs [2]. Today SVET still provides a unique ability to visualize the local current density of an active sample in electrolyte, providing a highly sensitive means to follow dynamic processes, like corrosion, as they occur.
How does the Scanning Vibrating Electrode Technique work?
The Scanning Vibrating Electrode Technique takes advantage of the potential gradient existing above a naturally active, or biased sample in electrolyte to map its local current density. Unlike predecessor techniques, like the Scanning Reference Electrode Technique (SRET), which mapped current density using a static probe, in SVET the probe is vibrated perpendicular to the sample resulting in a high signal-to-noise ratio.
Electrochemically active samples, like freely corroding samples, or biased samples produce a current above the surface when in solution. Due to iR drop a potential gradient exists in the electrolyte, with the conductivity of the electrolyte and strength of the sample activity affecting the final gradient. If a point in space is considered, the equipotential regions as illustrated in Fig. 1 arise. In the Scanning Vibrating Electrode Technique, the presence of the equipotential regions is exploited to perform the measurement. When the SVET probe is vibrated perpendicular to the sample it passes through different equipotential regions, meaning the potential measured is dependent on the position of the probe during its vibration. The result is the measurement of an ac potential with a dc offset. To produce the dc signal of interest, therefore, a Lock-In Amplifier is used to demodulate the ac signal. The dc potential can then be mapped relative to the probe position. This is similar to Local Electrochemical Impedance Spectroscopy (LEIS), which also utilizes the potential gradient arising from iR drop. In the case of LEIS, however, it is an ac signal of interest.
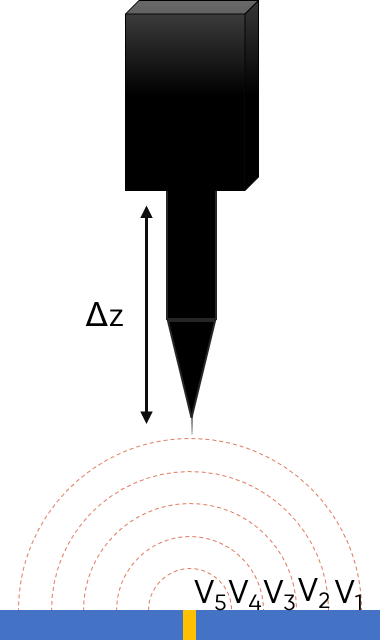
Figure 1: The working principle of the Scanning Vibrating Electrode Technique is illustrated for a point in space sample.
The value typically of interest when measuring a sample using the Scanning Vibrating Electrode Technique is the current density of the sample. In most cases, therefore, it is necessary to convert the measured potential to the current density via a simple calibration of the probe used in the measurement.
Potential and Current Distributions over a Disk Electrode with Vibrating Probe, a Mathematica demonstration of the principle of SVET may be of further interest.
What are the components used to perform the Scanning Vibrating Electrode Technique?

Figure 2: The components of a typical instrument capable of the Scanning Vibrating Electrode Technique are shown.
The components required to perform the Scanning Vibrating Electrode Technique are shown in the annotated image in Fig. 2. A SVET instrument is composed of an x,y,z scanning stage to allow the probe to precisely move into close proximity with the sample surface, and for x,y raster scanning to be performed. The control electronics act to interface between the various electronic components of the SVET instruments, and the control and measurement software, relating the measured signal to the probe position. The Scanning Vibrating Electrode Technique is dependent on the precision vibration of the probe perpendicular to the sample under study, a precision vibration stage in the form of a piezo is therefore necessary. When the Scanning Vibrating Electrode Technique is used the signal measured by the probe is high impedance, therefore an electrometer is required to amplify the resulting signal. In a SVET measurement the Lock-In Amplifier has two roles. The Lock-In Amplifier acts to generate the sine wave used during the probe vibration, it also acts to demodulate the ac signal measured by the probe to generate the dc voltage signal of interest. Finally, as with other scanning probe techniques the probe is of great importance in SVET. A typical SVET probe is an etched platinum tip which has been platinized. The platinization process acts to greatly increase the probe surface area, making it highly capacitive, and improving the signal measured.
Why use the Scanning Vibrating Electrode Technique?
The Scanning Vibrating Electrode Technique provides the unique ability to visualize local electrochemical processes of a naturally active sample by exploiting the local electrochemical current density in the solution above the sample, doing so in a non-invasive way. Furthermore, SVET is capable of achieving area scan measurements of a sample, due to its ability to use the sweep scan technique, in which the probe performs continuous measurements throughout each line. By vibrating the SVET probe the Scanning Vibrating Electrode Technique achieves a higher signal to noise ratio than its predecessor techniques, like the Scanning Reference Electrode Technique. All of this makes the use of the Scanning Vibrating Electrode Technique ideal for measuring dynamic samples, like those which are freely corroding, in situ. This is further benefitted by the sequencing of SVET measurements which allow changes over time to be mapped and followed.
What is the Scanning Vibrating Electrode Technique used for?
Figure 3: The uses of the Scanning Vibrating Electrode Technique are outlined. When applied to studies in biology SVET is referred to as vibrating probe.
As shown in Fig. 3 the Scanning Vibrating Electrode Technique has been applied in the fields of batteries, biology, coatings, and corrosion. Some examples of the use of the Scanning Vibrating Electrode Technique in these fields include:
- Following photosynthetic processes in real time [3]
- Following corrosion processes as they occur in situ [4]
- Studying the effect of sample structure on corrosion [5]
- Examining the self-healing ability of smart coatings [6]
- Investigating the effect of applied potential on aqueous battery electrode activity [7]
- Imaging electric fields associated with growth [8]
These uses of the Scanning Vibrating Electrode Technique are covered in detail in a series of Learning Center articles focusing on the research questions which can be answered using the various scanning probe electrochemistry techniques. For those areas of relevance to SVET please see:
Scanning probes & biology research
Scanning probes & coating research
Scanning probes & corrosion research
Further Information
To further your understanding of the Scanning Vibrating Electrode Technique, and other scanning probe electrochemistry techniques BioLogic offers a number of different resources including tutorials, application notes, and technical notes. And keep checking back on our Learning Center, which has new articles added regularly.
Glossary
- iR drop: Also known as ohmic drop, iR drop is the potential difference due to the passage of current, i, through a resistive component, i.e. the electrolyte, with resistance, R.
- Local Electrochemical Impedance Spectroscopy (LEIS): A technique related to SVET in which the local impedance of a sample is measured.
- Scanning Reference Electrode Technique (SRET): A predecessor to the Scanning Vibrating Electrode Technique. SRET maps the potential of a system through the use of a non-vibrating reference electrode. It has reduced sensitivity when compared to SVET.
- Vibrating probe: The term used instead of Scanning Vibrating Electrode Technique in the field of biology.
References
- F. Jaffe, Lionel F, R. Nuccitelli, J. Cell Biol. 63 (1974) 614-628
- S. Isaacs, J. Electrochem. Soc. 135 (1988) 2180-2183
- S. Lee, J. Plant Biol. 49 (2006) 186-192
- F. Glover, T. W. Cain, J. R. Scully, Corros. Sci. 149 (2019) 195-206
- Y. Liu, X. G. Li, Y. F. Cheng, Electrochem. Comm. 12 (2010) 936-938
- D. Wang, M. Y. Liu, J. J. Fu, J. Mater. Chem. A 3 (2015) 6423-6431
- Wang, Q. Wu, A. Abraham, P. J. West, L. M. Housel, G. Singh, N. Sadique, C. D. Quilty, D. Wu, E. S. Takeuchi, A. C. Marschilok, K. J. Takeuchi, J. Electrochem. Soc. 166 (2019) A3575-A3584
- Overall, L. F. Jaffe, Dev. Biol. 108 (1985) 102-119





