The 5 key points to optimize HPC analysis
Latest updated: April 1, 2026The importance of high-resolution instrumentation
High Precision Coulometry (HPC) has emerged as a transformative analysis in battery research, enabling scientists and engineers to quantify and optimize Coulombic Efficiency (CE) with unprecedented accuracy. CE has become a crucial diagnostic metric for understanding degradation mechanisms in lithium-ion batteries, where efficiencies routinely exceed 99.9% and where small differences can predict years of cycle life variation.
A useful metric for assessing the relative calendar life of a battery is its CE. When paired with measurements of internal cell resistance, high-precision CE testing provides a fast method for estimating the comparative lifetimes of different cell chemistries or designs. In general, CE is a time-dependent parameter that typically increases as the solid–electrolyte interphase (SEI) layers on both the negative and positive electrodes mature and their growth rate declines.
Achieving accurate CE measurement requires exceptional control of experimental conditions and high-resolution sensing across temperature, voltage, and current domains. BioLogic’s Premium Battery Cyclers, equipped with advanced analogue front ends, multi-range current measurements, high-resolution voltage sensing, millisecond or submilliseconds-scale time bases, and integrated temperature-control capabilities via the TCU Server, offer a uniquely powerful platform for this purpose.
This article outlines the scientific significance of CE measurement in lithium-ion batteries and explains how BioLogic’s hardware and control systems enable high-fidelity, reproducible CE analysis suitable for advanced research and industrial-quality validation, to ensure user benefits such as more reliable data, and time saving.
Introduction
Lithium-ion batteries underpin modern portable electronics, electric vehicles, grid storage systems, and emerging high-energy applications. As performance and longevity expectations rise, the ability to detect early decomposition pathways or subtle inefficiencies has become essential.
Coulombic Efficiency, defined as the ratio of discharge capacity to charge capacity, is one of the most sensitive indicators of long-term degradation in lithium-ion batteries.
$$CE = \frac{Charge\; delivered\; during\; discharge}{ Charge\; stored\; during\; the\; previous\; charge} x 100$$
Small changes in CE often signal the onset of side reactions such as solid electrolyte interphase (SEI) thickening, electrolyte oxidation, and lithium plating. Because the magnitude of these changes is extremely small, a typical lithium-ion cell may vary by only 0.01% in CE between stable and degrading cycling conditions.
Coulombic Efficiency reflects the reversibility of electrochemical reactions occurring during cycling. In an ideal lithium-ion battery, all the charge inserted during charging would be extracted during discharge. However, real cells exhibit parasitic side reactions that consume charge. Over time, these losses manifest as reduced cyclable lithium inventory, impedance growth, and capacity fade. Because these parasitic currents are often extremely small (sometimes in the range of microamps over extended CV holds) detecting them requires instrumentation that minimizes noise, offset, and thermal drift.
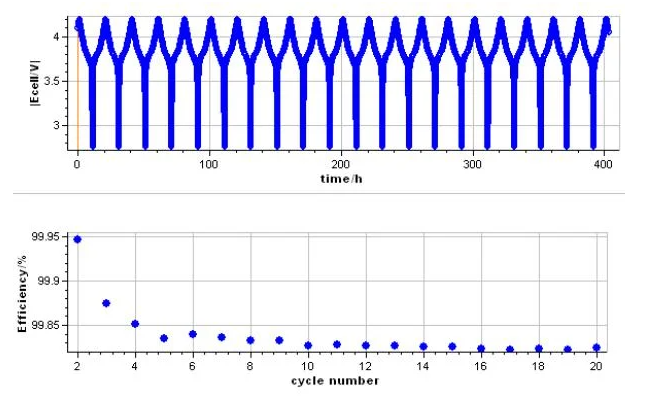
A CE value of 99.95% may appear robust but can indicate drastically different long-term performance from a value of 99.99%. The difference corresponds to cumulative losses that dramatically shorten the lifetime of high-energy cells, especially those using nickel-rich cathodes or silicon-dominant anodes. High-fidelity CE measurement is therefore fundamental not only for academic research into degradation mechanisms but also for industrial tasks such as evaluating additives, assessing formation protocols, and comparing novel electrode formulations.
These variations can only be identified through high-resolution instrumentation capable of controlling and measuring the battery environment with scientific precision. Accurate CE tracking demands strict control of temperature, precise voltage cutoffs, Ohmic drop correction, high-resolution current measurements, and accurate time integration—conditions that BioLogic systems are designed to fulfil.
BioLogic potentiostat and battery cyclers, known for their 18-bit analog-to-digital conversion, millisecond or submilliseconds-level time base, five selectable current ranges, and integrated TCU Server for temperature-chamber control, provide a tightly engineered platform suitable for high-resolution Coulombic Efficiency measurement. Over the following sections, this paper explores how measurement resolution, thermal conditions, current-range selection, and system-level synchronization contribute to accurate CE quantification.
1. Thermal Control and Temperature Resolution
Temperature stability is the most important environmental factor affecting lithium-ion cell behaviour. Reaction kinetics for SEI growth, electrolyte decomposition, and metal deposition follow Arrhenius-type relationships, making them highly sensitive to minor fluctuations in temperature. Even small drifts less than 1°C can produce measurable changes in side reaction rates, which then appear in CE data. Achieving meaningful CE measurement therefore requires both high thermal stability and high resolution.
BioLogic systems integrate temperature chamber control through the TCU Server, which provides direct integration between the instrument and a programmable thermal chamber, providing a closed feedback loop for temperature regulation.
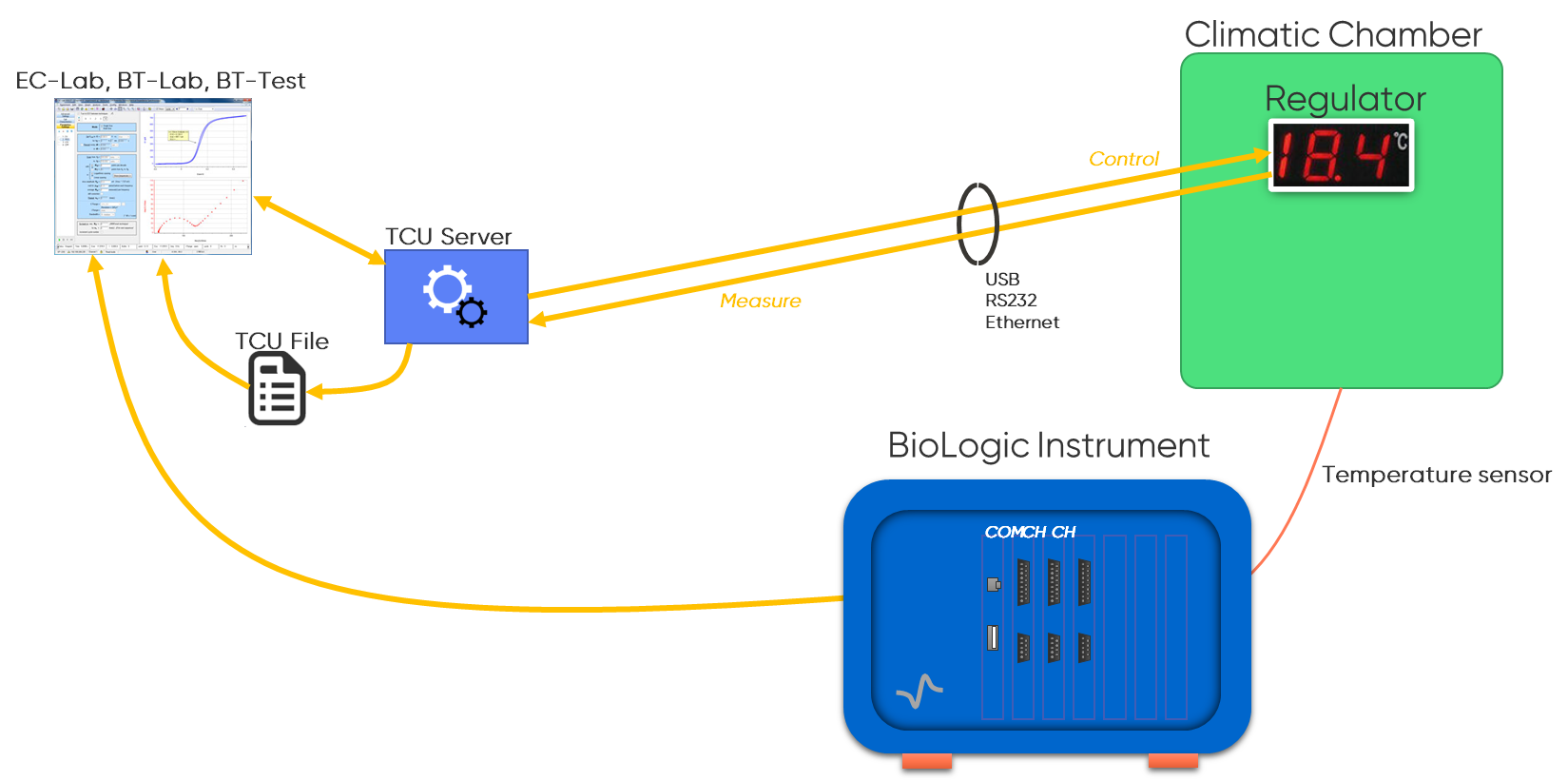
This integration ensures that the chamber can react to and control temperature with fine resolution and long-term stability. With continuous bidirectional communication, the system maintains regulation of the battery’s thermal environment throughout cycling. This allows the identification of genuine CE changes rather than artifacts produced by thermal drift. Because lithium plating thresholds, high-voltage electrolyte oxidation, and impedance profiles are temperature dependent, stable temperature control is essential to avoid misinterpreting thermal effects as electrochemical degradation.
2. Voltage Resolution and Cutoff Accuracy
Voltage resolution is critical in lithium-ion CE measurement due to the non-linear shape of the open-circuit-voltage curve, particularly near high state of charge. BioLogic cyclers incorporate high-resolution 18-bit analog-to-digital conversion, which over a typical 5 V range yields microvolt-scale resolution. Such fine measurement precision ensures that charge and discharge cut-offs occur at exactly the intended voltage each cycle. In high-precision experiments, voltage deviations as small as 1–2 mV can introduce quantifiable errors in measured capacity because small voltage differences correspond to meaningful movements along the State of Charge (SOC) curve.
During CV charging, current may decline toward extremely small values, and any offset in the measurement system will accumulate into a charge integration error over time. BioLogic’s low-noise amplifier architecture and range-switching logic minimize such offsets, ensuring smooth transitions between measurement ranges without generating discontinuities. The ability to switch ranges without compromising accuracy is especially important for Li-ion studies involving low-rate cycling used for high-precision CE measurement. Combined with millisecond-level time-base data acquisition, BioLogic’s multi-range design prevents loss of detail in current transients and improves the accuracy of capacity integration.
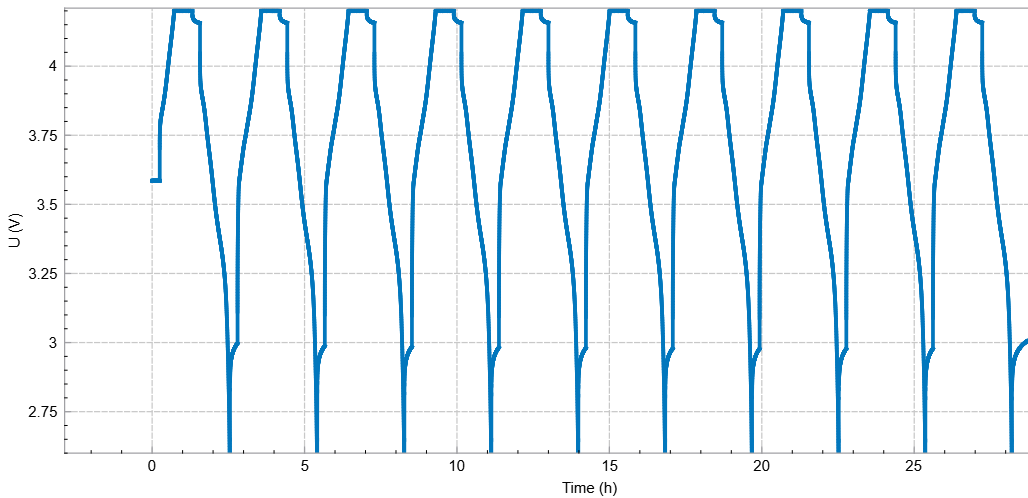
Accurate cutoff control also prevents excursions into voltage regimes that accelerate degradation. For instance, at high SOC near 4.2 V, even slight overcharging may intensify electrolyte oxidation and lattice instability in nickel-rich cathodes. High-resolution voltage sensing allows the system to terminate charge precisely before entering the region where parasitic reactions become dominant. Thus, ensures that apparent capacity variations reflect genuine electrochemical phenomena rather than instrumental artifacts. The stability and repeatability of BioLogic’s voltage cutoff system thus form a core component of reliable CE measurement.
3. Ohmic drop correction
Ohmic drop (or IR drop) refers to the potential error caused by the resistance of the electrolyte, connectors, or other resistive components in an electrochemical cell. This resistance creates a voltage drop when current flows, which can distort your measurements—especially in techniques like cyclic voltammetry, impedance spectroscopy, or corrosion studies. When ohmic drop is present, the measured voltage may be higher or lower than the true electrode potential, leading to incorrect cut-off voltages during charge/discharge cycles. This can result in overcharging, undercharging, or miscalculated capacity values, all of which distort CE calculations.
Ohmic correction is the process of compensating for this unwanted voltage drop, ensuring that the measured potential more accurately reflects the true electrochemical processes at the electrode surface. The ability to correct for ohmic drop is particularly important for accurate CE calculations. Uncompensated ohmic drop can lead to errors in voltage and current measurements, directly impacting the calculated charge and discharge capacities—and thus the CE.
Potentiostats
BioLogic potentiostats, integrated with EC-Lab® software, offer robust support for ohmic drop compensation, improving the accuracy of CE measurements, especially in battery testing and electrochemical research. On the hardware side, BioLogic potentiostats are capable of compensating for ohmic drop, though this is constrained by the current range’s shunt resistor—such as a 100 Ω limit for a 10 mA range.
For ohmic drops exceeding these hardware limits, EC-Lab® provides powerful software-based solutions. The software includes multiple techniques for determining and correcting ohmic drop, such as:
- Current interrupt – measures the instantaneous voltage change upon current interruption to calculate solution resistance
- Electrochemical Impedance Spectroscopy (EIS) – uses impedance data to estimate and compensate for ohmic resistance
- ZIR (Zero Impedance Resistance) – single-frequency impedance method for precise ohmic drop determination.
Additionally, users can seamlessly apply ohmic drop compensation in linked experiments and fine-tune parameters directly within the EC-Lab® environment, ensuring both flexibility and accuracy in their electrochemical analyses
Battery Cyclers
In the case of BCSs, BT-Lab Suite® integrates techniques like Electrochemical Impedance Spectroscopy (EIS) to measure and correct ohmic drop in real time. Thanks to the variables in the CALCULATE Task, you can apply ohmic drop correction across linked experiments without manual intervention.
This ensures that voltage readings reflect the true electrochemical processes at the electrode, not resistive losses.
By applying ohmic drop correction, BioLogic instruments ensure that the voltage and current measurements are more accurate and reproducible, allowing for precise determination of charge and discharge capacities. This leads to more reliable CE values, which are essential for evaluating battery performance, degradation mechanisms, and cycle life.
To know more, check out: https://www.biologic.net/topics/ohmic-drop-correction-a-means-of-improving-measurement-accuracy-with-potentiostats/ ; https://www.biologic.net/documents/ohmic-drop-zir-techniques-electrochemistry-battery-application-note-29/ and https://www.biologic.net/documents/ohmic-drop-measurement-techniques-electrochemistry-battery-application-note-28/
4. Current Measurement Across Multiple Ranges
The accuracy of Coulombic efficiency ultimately depends on the ability to measure current with high precision, because CE is calculated at each time base as it is processed by the firmware (not sampling dependent). Check out https://www.biologic.net/topics/data-of-interest-getting-the-most-out-of-your-blue-box/ to know more.
BioLogic instruments provide five independent current ranges, allowing the system to operate near the optimal point of the measurement electronics for widely differing cell chemistries and capacities. This ensures an optimal signal-to-noise ratio regardless of whether a cell is operated at microampere-scale research currents or amp-level cycling conditions. Because many parasitic reactions occur at microamp-level magnitudes, the low current ranges are essential for identifying the inefficiencies that give rise to CE loss.
Selecting the correct current range minimizes measurement noise and maximizes dynamic resolution. Select the appropriate instrument configuration: BCS instruments exist with different current capability and have up to five current ranges each; Potentiostat can be optimized depending on your needs.
5. Importance of Millisecond-Level Time Base
Time precision is often overlooked in CE measurement, yet it plays a critical role because the total charge is the integral of current over time. BioLogic’s millisecond time base is not sampling dependent as it is managed by the firmware, and it ensures that every transient event—such as the voltage knee near cutoff, stabilization during rest periods, or short-lived current disturbances—is properly captured. Missing or approximating these fast events introduces numerical integration errors, which accumulate over the duration of a full charge–discharge cycle.
In lithium-ion experiments where CE shifts are sometimes only a fraction of a milliamp-hour per cycle and these extremely small timing inaccuracies can become significant. BioLogic’s fast-logging rate ensures that current integration uses a finely sampled dataset, minimizing numerical drift. This increased temporal fidelity is critical because Coulombic Efficiency values often differ by less than 0.1%. Precise time resolution ensures that capacity measurements reflect true electrochemical processes, such as lithium intercalation kinetics and parasitic reactions, rather than artifacts from data under sampling or interpolation. The ability of the BioLogic system to resolve transient signals and maintain accurate time synchronization ensures that cycle-by-cycle CE calculations remain stable and reflect the actual electrochemical conditions within the cell.
Combined Impact on Coulombic Efficiency Analysis
Temperature stability, high voltage resolution, precision current measurement, and accurate time integration collectively determine the reliability of CE measurements. BioLogic’s integrated approach ensures that each of these factors works in harmony. The TCU Server maintains a stable environment; the 18-bit voltage measurement system ensures precise and repeatable cutoff behaviour; the multiple current ranges allow accurate sensing of both large and small currents; and the millisecond time base ensures exact charge integration.
When combined, these features create a measurement infrastructure where changes in CE can be directly attributed to electrochemical processes rather than experimental artifacts. Such fidelity is essential for research into next-generation lithium-ion systems, including high-nickel cathodes, silicon-dominant anodes, lithium-metal hybrids, and electrolyte additive packages designed to suppress SEI growth or plating. HPC analysis using BioLogic systems enables precise assessment of these technologies and supports the development of batteries with longer life, higher safety margins, and improved energy density by measuring Coulombic Efficiency.
References
- Shunli Wang, Yongcun Fan, Daniel-Ioan Stroe, Carlos Fernandez, Chunmei Yu, Wen Cao, Zonghai Chen,Battery System Modeling,Elsevier,2021
- Balazs Gyenes, D. A. Stevens, V. L. Chevrier and J. R. Dahn, Understanding Anomalous Behavior in Coulombic Efficiency Measurements on Li-Ion Batteries, Journal of The Electrochemical Society, Volume 162, Number 3Citation Balazs Gyenes et al 2015 J. Electrochem. Soc. 162 A278DOI 10.1149/2.0191503jes
- Attia, P. M., Bills, A., Brosa Planella, F., de Soto, F., van Vliet, D., Yang, Z., & Bazant, M. Z. (2020). Closed-loop optimization of fast-charging protocols for batteries with machine learning. Nature, 578, 397–402. https://doi.org/10.1038/s41586-020-1994-5
- BioLogic Science Instruments. https://www.biologic.net
- Biot, M., Gauthier, M., & Mohtadi, R. (2022). Electrochemical characterization techniques for advanced battery materials. Advanced Energy Materials, 12(6), 2102566. https://doi.org/10.1002/aenm.202102566
- Delacourt, C., & Safari, M. (2011). Mathematical modeling of lithium batteries: From solid-state physics to practical models. Journal of The Electrochemical Society, 158(10), A1143–A1160. https://doi.org/10.1149/2.051111jes
- Ecker, M., Nieto, N., Käbitz, S., Schmalstieg, J., Bläske, F., Müller, J., & Sauer, D. U. (2014). Calendar and cycle life study of Li(NiMnCo)O₂-based 18650 lithium-ion batteries. Journal of Power Sources, 248, 839–851. https://doi.org/10.1016/j.jpowsour.2013.09.143
- Noh, H., Yoon, C. C., Lee, E., Kim, S., & Cho, J. (2020). Advanced diagnostic methods for lithium-ion batteries. Nature Communications, 11, 6158. https://doi.org/10.1038/s41467-020-19954-z
- Olivier, L., & Bultel, Y. (2018). High-precision coulometry for battery degradation studies. Electrochimica Acta, 274, 1–12. https://doi.org/10.1016/j.electacta.2018.04.119





