Scanning Droplet Cell with ICP-MS
Latest updated: February 6, 2026Scanning Droplet Cell (SDC) is used to perform direct local electrochemistry measurements of a sample through the formation of a droplet between a specially designed head or glass capillary and the sample, which acts as a working electrode. Using SDC, it is possible to measure the local activity of a sample, while only exposing a small region of the sample to electrolyte at a time. This has seen SDC becoming increasingly popular for both corrosion and catalysis studies, in particular for high throughput screening of materials of interest. With high throughput screening, it can be beneficial to measure multiple characteristics of a sample within one workflow. Keeping that in mind, it is appealing to measure more than just activity when using SDC for library screening and is the goal when using SDC with inline analysis. Of growing interest is the use of SDC with Inductively Coupled Plasma – Mass Spectrometry (ICP-MS), allowing high throughput screening of both activity and stability.
What is ICP-MS and why is it used with SDC?
ICP-MS, or Inductively Coupled Plasma – Mass Spectrometry, is a form of mass spectrometry which uses plasma to ionize the sample of interest, typically a liquid, ready for measurement by mass spectrometry [1, 2]. It is used to measure trace elements, with a high level of sensitivity, as low as Parts Per Trillion (PPT) [3, 4].
The high sensitivity of ICP-MS to trace elements makes it highly useful to couple with SDC, providing a means to measure both activity and stability at once [5]. ICP-MS has further advantages including the ability to measure time resolved dissolution information [6], detect multiple elements simultaneously [5], and distinguish between different isotopes [7]. These traits make the coupling of ICP-MS with SDC particularly useful for studies of catalysis and corrosion materials.
How is SDC coupled to ICP-MS?
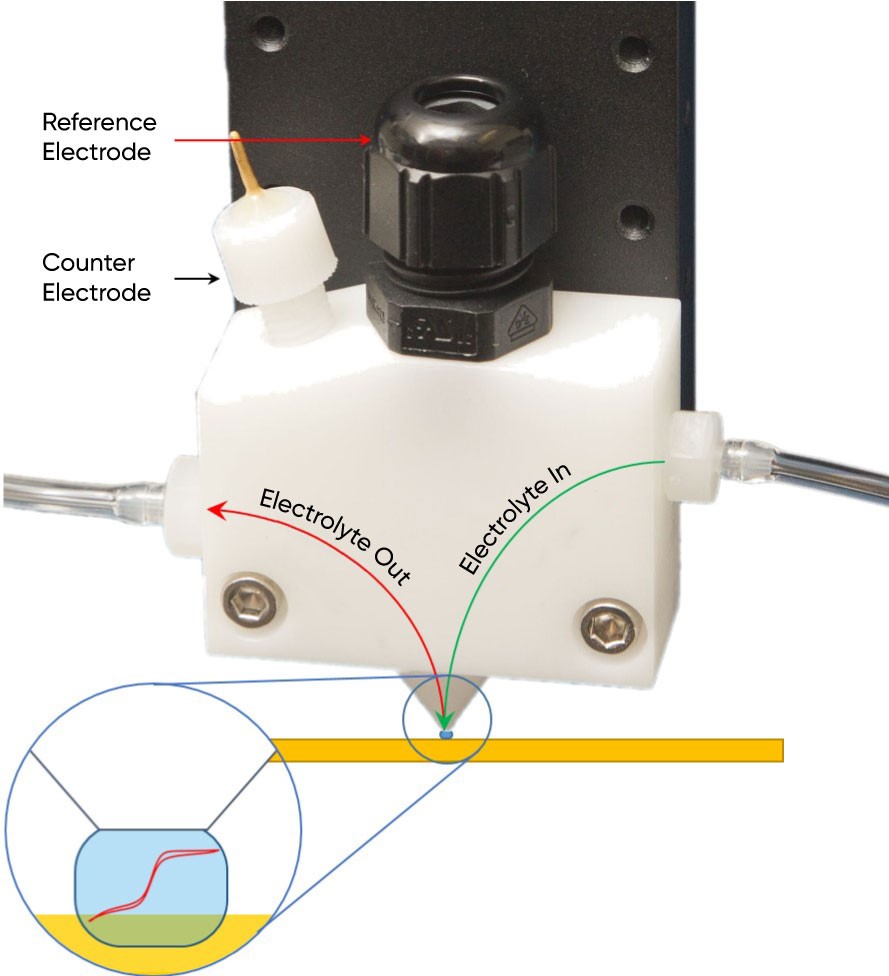
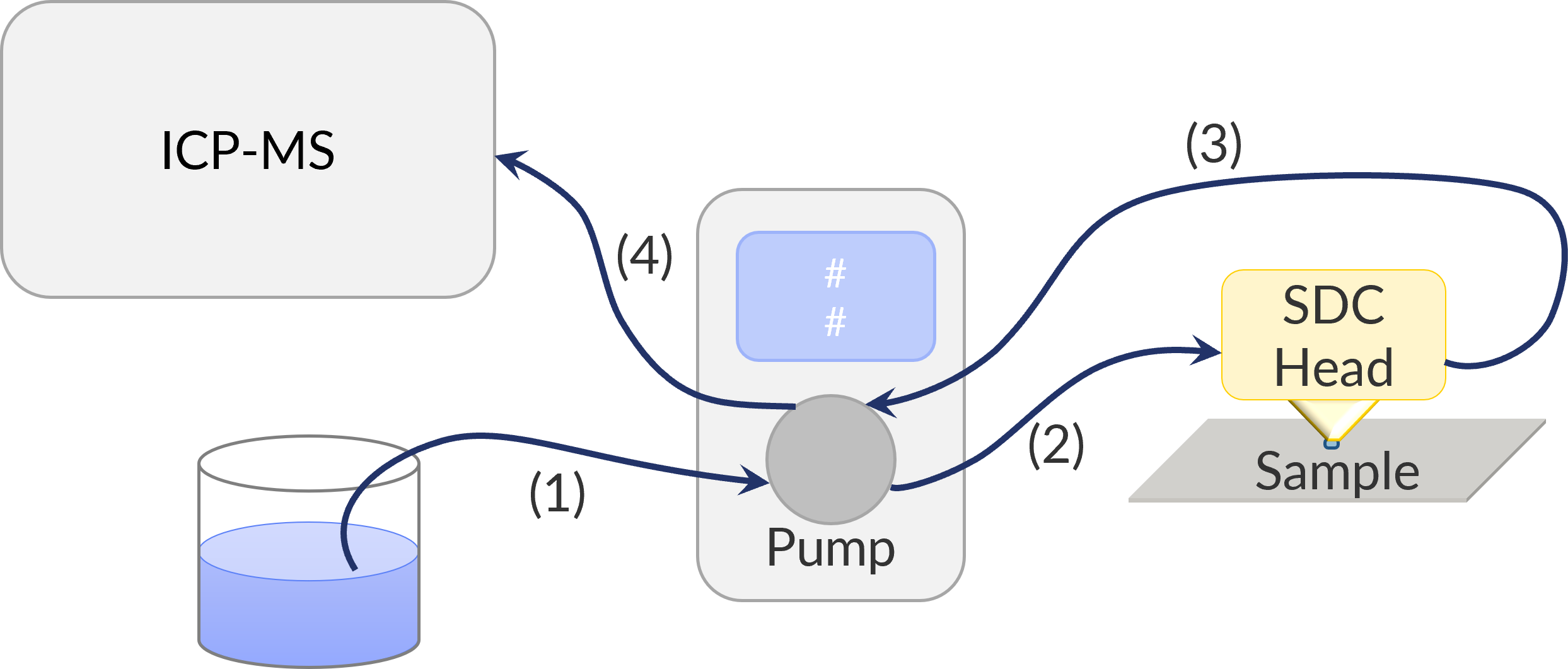
When ICP-MS is coupled to SDC it is used as a downstream analysis, meaning the local electrochemistry experiment of SDC is first and then ICP-MS is performed. This means ICP-MS must be coupled with a flow type SDC head, Fig. 1, where the electrolyte flows into the SDC head to the droplet, and then out of the measurement head. Because of the electrolyte flow, after the SDC experiment, any species which have dissolved into the electrolyte then flow through to the ICP-MS for the mass spectrometry experiments to be performed. This is illustrated in Fig. 2.
Figure 1 : The SDC flow head is shown, with the electrodes, and direction of electrolyte flow shown.
Figure 2 : The flow of electrolyte through the SDC coupled with ICP-MS experiment is shown. The electrolyte first flows from its source to the peristaltic pump (1), from the peristaltic pump it then flows into the SDC head to form the droplet (2), from the SDC head it then flows back to the peristaltic pump, through a second channel (3), finally from the peristaltic pump it flows to the ICP-MS (4). Note components are not drawn to scale.
When coupling SDC and ICP-MS there are a few points that must be kept in mind.
- While the different stages can be performed manually, for large libraries, a means to automate the process can be beneficial. This is possible using the sequencer of BioLogic’s M470, with the interaction with a connected device possible with the USB-PIO.
- The flow rate used is important. If the flow rate is too fast there can be difficulties in the measurement and data interpretation, due to the build up of bubbles and chemical species [8].
- The tubing length between the SDC head and ICP-MS is important. Short tubes are generally better because they have a shorter dead time and offer better separation of dissolution peaks during the SDC test [4]. It is important to also ensure consistency in tube length between experiments. [8]
Conclusion
Coupling of SDC with ICP-MS offers a means of advancing the data which can be acquired from SDC experiments, with the electrochemical activity and stability of a sample measured in one workflow. This is particularly beneficial when considering the growing interest in high throughput screening of materials for catalysis and corrosion.
References
- https://en.wikipedia.org/wiki/Inductively_coupled_plasma_mass_spectrometry
- A. Sader, S. Ryan, Geochemistry: Exploration, Environment, Analysis 20 (2019) 167-175
- E. Adesina, C. J. Burgos, T. R. Grier, A. S. M. Sayam, A. J. Specht, Current Environmental Health Reports 12 (2025) 7
- Moriau, T. Đukić, V. Domin, R. Kodym, M. Prokop, K. Bouzek, M. Gatalo, Martin Šala, N. Hodnik, Electrochimica Acta 487 (2024) 144200
- J. Jenewein, G. D. Akkoc, A. Kormányos, S. Cherevko, Chem Catalysis 2 (2022) 2778–2794
- Ooi, E. Tada, A. Nishikata, Materials Transactions 62 (2021) 797- 806
- W. Hassel, M. Hafner, Physica Status Solidi A 219 (2022) 2200029
- Shkirskiy, F. D. Speck, N. Kulyk, S. Cherevko, Journal of The Electrochemical Society, 166 (2019) H866