LEIS101: An Introduction to Local Electrochemical Impedance Spectroscopy
Latest updated: November 18, 2024What is Local Electrochemical Impedance Spectroscopy?
Local Electrochemical Impedance Spectroscopy (LEIS) builds on the principles of Electrochemical Impedance Spectroscopy (EIS) to measure the local impedance of a sample. LEIS can be performed at a single frequency to map the local impedance of a sample, or the probe can be held stationary while varying the ac frequency to measure a full Electrochemical Impedance Spectrum at a single point.
Local Electrochemical Impedance Spectroscopy was introduced in 1992 by Hugh Isaacs to measure the local impedance characteristics of a sample [1]. Though EIS has been extensively used in bulk analysis the resulting data is an average of the entire surface. If the impedance of a sample is heterogeneous, as would occur for a sample with local corrosion, it can be difficult to interpret the resulting EIS data to determine the contribution from different local features. LEIS, on the other hand, allows the EIS of individual features to be measured independent of the bulk sample, and furthermore can be used to visualize these impedence variations across a sample. As one of the only techniques which allows local impedance data LEIS has carved a niche in studies where the local characteristics are of interest, finding use in corrosion and coatings studies.
How does Local Electrochemical Impedance Spectroscopy work?
Local Electrochemical Impedance Spectroscopy works on the principle that the local impedance of a sample affects the ac current response in solution, which can be measured by a probe in close proximity to the sample. In LEIS, a sinusoidal voltage or current perturbation is applied to a sample in solution, in much the same manner as in bulk EIS measurements. As a result, an ac current flows through the solution, and a potential gradient develops above the sample. LEIS uses a bi-electrode probe to measure ac potential difference, which ultimately allows the local impedance of the sample to be determined, Fig. 1.
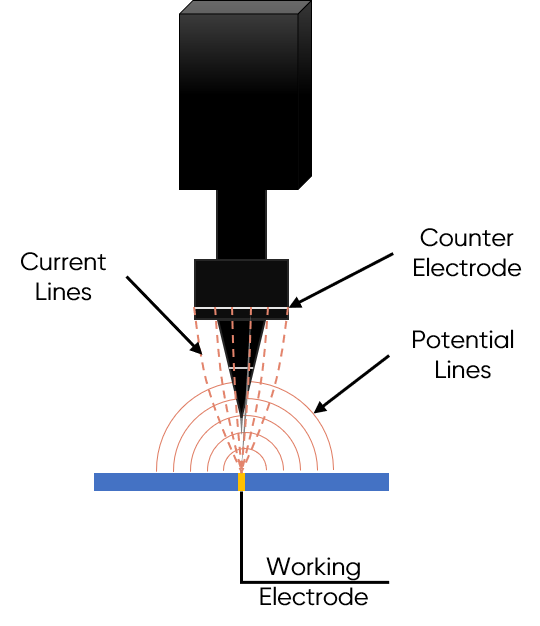
Figure 1: The working principle of Local Electrochemical Impedance Spectroscopy is illustrated.
Local Electrochemical Impedance Spectroscopy uses a five-electrode cell. The standard three-electrode cell in which the sample is the working electrode, composes part of this. An ac voltage is applied between the working electrode sample and the reference electrode, resulting in current flow between the sample and the counter electrode. The final two electrodes of the cell are those of the bi-electrode LEIS probe. The bi-electrode probe allows the ohmic drop in solution parallel to the sample surface to be exploited. The two electrodes of the LEIS probe have a known height difference between them. Because the ac potential varies with distance from the sample surface, having the electrodes of the probe at different heights results in a potential difference between them. This potential or voltage difference is measured using an electrometer and analyzed to obtain the sample local impedance.
There are two main experiment types which can be performed in LEIS: (1) line/area scan and (2) frequency sweep. LEIS line and area scans, sometimes referred to as Local Electrochemical Impedance Mapping (LEIM), are performed at a single frequency while the probe is raster scanned. These measurements provide a map of the local impedance data. These maps would typically be the first experiment performed to pinpoint areas of interest before moving to frequency sweep measurements. Frequency sweep measurements are the Electrochemical Impedance Spectroscopy measurements performed at a stationary x,y point in close proximity to the sample surface. As the name implies these measurements are performed over a range in frequencies, allowing the Nyquist and Bode plots users will be familiar with from bulk EIS to be acquired.
What are the components of a Local Electrochemical Impedance Spectroscopy instrument?

Figure 2: Annotated image of an instrument capable of performing Local Electrochemical Impedance Spectroscopy.
The components of an instrument capable of performing Local Electrochemical Impedance Spectroscopy are annotated in Fig. 2. As with other scanning probe electrochemistry techniques the control electronics act to interface between the software and all of the electronic devices of the instrument. The x,y,z scanning stage is used to approach the probe into close proximity with the sample, and to perform automated raster scanning in x and y. In LEIS a single combined potentiostat/galvanostat and Frequency Response Analyzer (FRA) is used, this is connected to the sample which acts as the working electrode throughout the measurement. The potentiostat (galvanostat) and FRA performs the impedance measurement, at a single frequency or over a range of frequencies, while also controlling the potential (current). The electrometer measures the potential difference at the bi-electrode probe used in LEIS, while the Lock-In Amplifier demodulates the signal measured at the probe. A bi-electrode probe is used in LEIS as described to allow a potential difference in solution to be measured. The probe affects the final resolution of the measurement. However, it is important to note that while larger probes result in a lower resolution measurement, they allow an increased frequency range to be studied. Finally, it is important to mention the counter electrode used in the LEIS measurement, which can affect the final signal measured. When performing LEIS measurements, it is important to select a counter electrode with axial symmetry about the LEIS probe. Such a configuration ensures current lines from the sample are perpendicular to the plane of the sample, and that features measured correlate to physical features. Using different counter electrode configurations can lead to distorted images, and/or artefacts, though this effect can disappear when the probe is close to the sample surface.
Why use Local Electrochemical Impedance Spectroscopy?
Bulk EIS has been applied to study a wide variety of sample types due to its high sensitivity to the electrochemical processes at the electrode interface. EIS is limited, however, in only providing an average measurement of the sample under study. When there is variation in the electrochemical activity across a sample EIS can be difficult to interpret, this is where Local Electrochemical Impedance Spectroscopy comes in. By measuring only, the region under the electrode LEIS provides truly local information about the sample impedance allowing the influence of features on the overall sample impedance ot be directly measured. LEIS is one of only a small number of techniques capable of measuring the local impedance of a sample, the others being ac-Scanning Electrochemical Microscopy, and ac-Scanning Droplet Cell. Using LEIS, it is possible to perform high sensitivity measurements to obtain local electrochemical data about the sample of interest in a non-destructive manner. Local Electrochemical Impedance Spectroscopy has the particular advantage that it can even be used in the study of coated samples. Its high sensitivity has been demonstrated in this respect with LEIS capable of detecting defects in surface coatings before they are visible.
What is Local Electrochemical Impedance Spectroscopy used for?
Figure 3: The uses of Local Electrochemical Impedance Spectroscopy are outlined.
Local Electrochemical Impedance Spectroscopy has been used extensively for the related fields of coatings and corrosions research, and to a lesser extent in battery investigations, Fig. 3. Some examples of the use of Local Electrochemical Impedance Spectroscopy include:
• Studying the ability of smart coatings to self-heal [2]
• Investigating the occurrence of underfilm corrosion [3]
• Examining the effect of alloy content on corrosion [4]
• Investigate the effect of external stress on corrosion [5]
• Investigating the effects of surface treatments on battery electrodes [6]
For more detailed information on the use of LEIS and other scanning probe electrochemistry techniques in these applications please see:
Scanning probes & coating research
Scanning probes & corrosion research
Scanning probes & battery research
Further Information
BioLogic offers a wide variety of tutorials, application notes, and technical notes to expand your understanding of the techniques we offer. And keep checking back on our Learning Center, which we update regularly!
Glossary
Electrochemical Impedance Spectroscopy (EIS): A bulk electrochemical measurement in which a sinusoidal bias is applied to a sample over a set frequency range to obtain information about electrochemical processes .
iR drop: Also known as ohmic drop, iR drop is the potential difference due to the passage of current, i, through a resistive component, i.e. the electrolyte, with resistance, R.
Local Electrochemical Impedance Mapping (LEIM): The name sometimes used to refer to LEIS raster scanning measurements performed at a single ac frequency.
References
- S. Lillard, P. J. Moran, H. S. Isaacs, J. Electrochem. Soc. 139 (1992) 1007-1012
- R.Raj, Y. Morozov, L. M. Calado, M. G. Taryba, R. Kahraman, A. Shakoor, M. F. Montemor, Electrochim. Acta 319 (2019) 801-812
- V. S. Philippe, G. W. Walter, S. B. Lyon, J. Electrochem. Soc. 150 (2003) B111-B119
- T. Liu, Y. Wang, S. Pan, Q. Zhao, C. Zhang, S. Gao, Z. Guo, N. Guo, W. Sand, X. Chang, L. Dong, Y. Yin, Corros. Sci. 149 (2019) 153-163
- A. Zhang, Y. F. Cheng, Corros. Sci. 52 (2010) 960–968
- L. Wang, L. M. Housel, D. C. Bock, A. Abraham, M. R. Dunkin, A. H. McCarthy, Q. Wu, A. Kiss, J. Thieme, E. S. Takeuchi, A. C. Marschilok, K. J. Takeuchi, ACS Appl. Mater. Interfaces 11 (2019) 19920-19932





